
Partnering with developing countries to improve brain health worldwide
The Ohio State University’s Neurological Institute is the highest nationally ranked program for neurology and neurosurgery in Columbus with over 200 medical, surgical and research specialists dedicated to the treatment of patients with neurological and psychiatric disorders.
Read more about our latest clinical trials and research innovations taking place every day. Visit the Neurology and Neurosurgery Department websites for additional information.

New Division of Neurogenetics paves way for expanded personalized medicine through clinical trials, gene targeted therapies
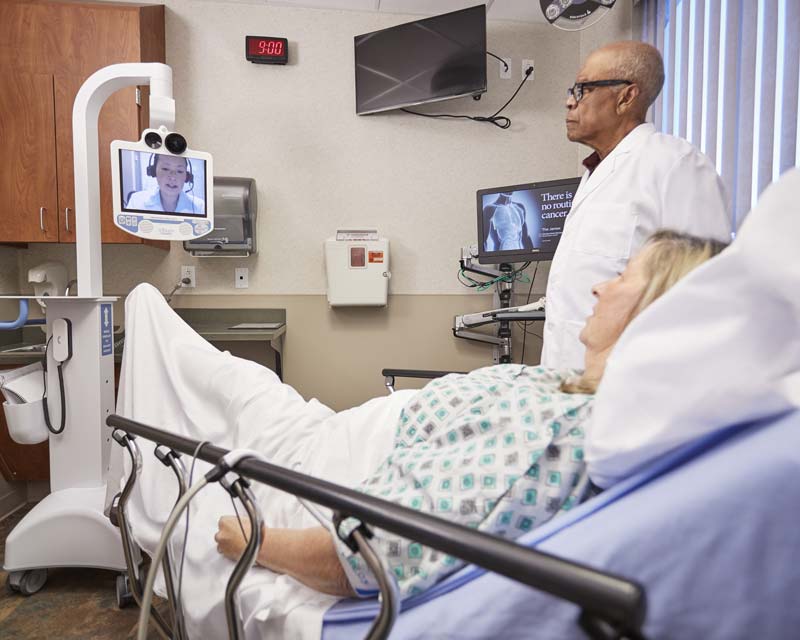
Neurology physician implementing teleneurology services to connect academic medical providers with local physicians

The Department of Neurology launches Division of Health Services Research

Neurology physician committed to educating tomorrow’s multiple sclerosis specialists
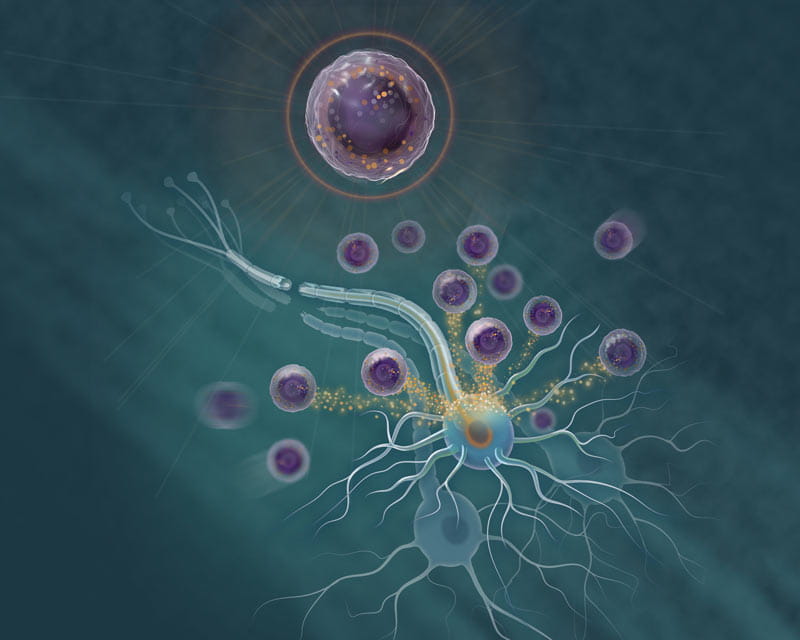
Neurology researchers testing novel immune cell’s ability to rescue damaged human nerve cells and induce the nerve fiber regeneration

Neurology researchers receive NIH grant, study what causes loss of motor function as we age
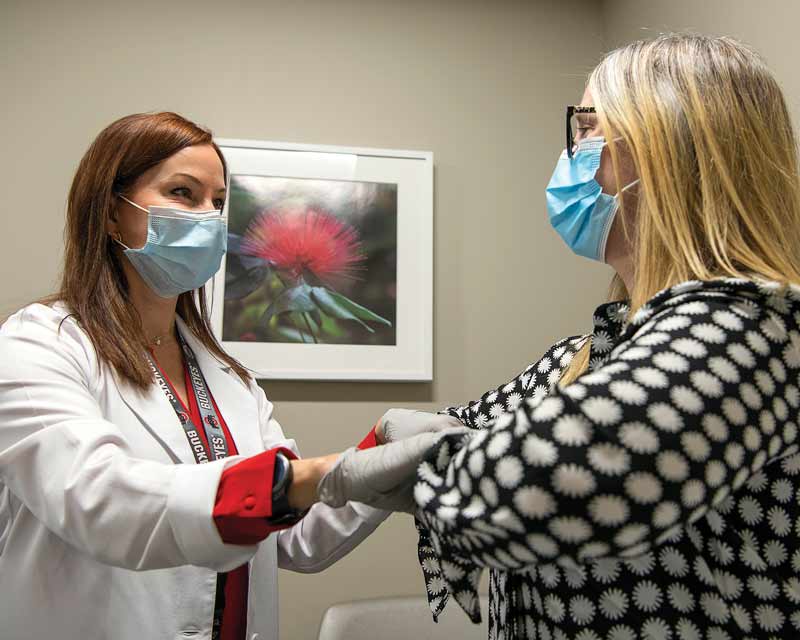
New specialty clinics offer highly personalized care to patients with neurological disorders
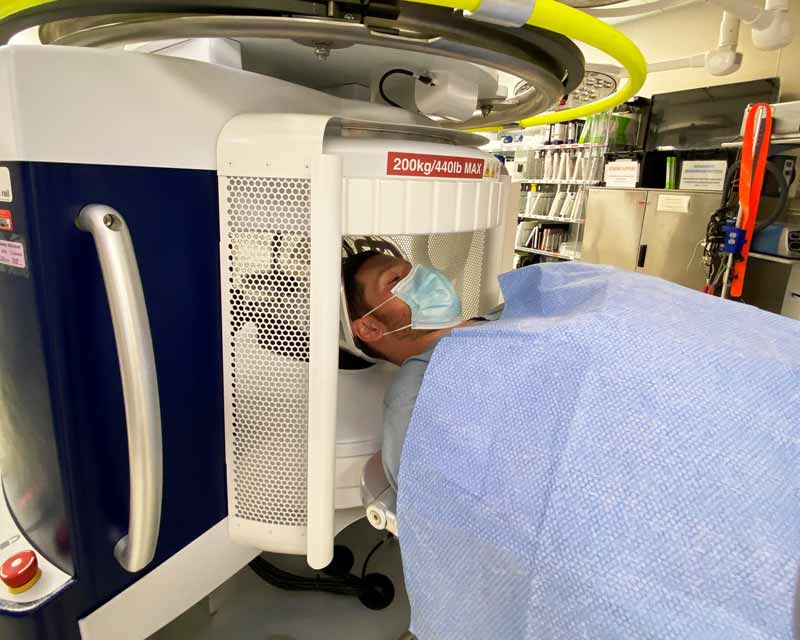
Portable MRIs provide lifesaving bedside brain imaging for stroke patients

Brain bank expansion will boost novel neurological research

Recent Department of Neurology expansion includes advances in teleneurology, accessibility, and genetics and aging research
A new approach to gene therapy provides hope to those living with incurable neurologic disorders

Directional and sensing DBS system delivers more personalized treatment for Parkinson’s disease
Certain immune-suppressing therapies reduce vaccine response in subset of multiple sclerosis patients

Novel research explores neurobiological mechanisms driving sarcopenia in aging

New partnership brings SAGE to a digital format

Ohio State establishing national standards for ALS genetic testing, research and patient care
Ohio State researchers identify new biomarkers that differentiate Lewy body dementia from Alzheimer’s and Parkinson’s

Biomarker breakthroughs at Ohio State transform disease diagnosis, predictability and treatment
The Ohio State University Wexner Medical Center pushes boundaries for neurological care

Multiple sclerosis and COVID-19: Experts at The Ohio State University Wexner Medical Center use data to guide care
Creating quantifiable metrics for back pain

Discovery of Pro-Regenerative Neutrophils Could be Breakthrough in Treating Incurable Neurological Diseases

Exploring a Onetime Treatment to Halt Parkinson’s Disease

Promising Clinical Trial Shows Benefit of Neuroprotective Therapy in Patients with ALS

The Past, Present and Future of Gene Therapy for Spinal Muscular Atrophy and Neuromuscular Diseases

New Approaches to Treating Movement Disorders with Deep Brain Stimulation and Other Advanced Treatments

Ohio State Researchers Study How to Use the Immune System to Promote Repair in Individuals with Neurological Disease

Krystof Bankiewicz, MD, PhD, Brings Promising Gene Therapy Research to Ohio State

New Neurology Chair Shares Vision for Growth

New Research Center at Ohio State to Study Neuroinflammation

Ohio State Pioneers Ultrasound Technology for Neurological Disorders

Ohio State Researchers Lead Spinal Cord Injury Center

Ohio State Wexner Medical Center Among Nation's Best in 4 Specialty Areas