May 14, 2018
(PLEASE NOTE: This clinical trial has ended.)
COLUMBUS, Ohio – Researchers at The Ohio State University Wexner Medical Center are exploring how small changes in the nasal cavity impact airflow and quality of life.
As part of a clinical trial, doctors use a new, non-invasive approach to reshape nasal tissue. The Vivaer Nasal Airway Remodeling device delivers radiofrequency energy to the nasal valve area to treat nasal obstruction, a condition that impacts millions of Americans.
“What this technology does is reshape the internal nasal valve region, which is a region where cartilage on the side of your nose meets your septum,” said Dr. Brad Otto, assistant professor of otolaryngology at Ohio State’s Wexner Medical Center. “Basically what it causes the cartilage to do is barely denature and change its shape just a little bit in order to open up that valve and improve airflow to that region.”
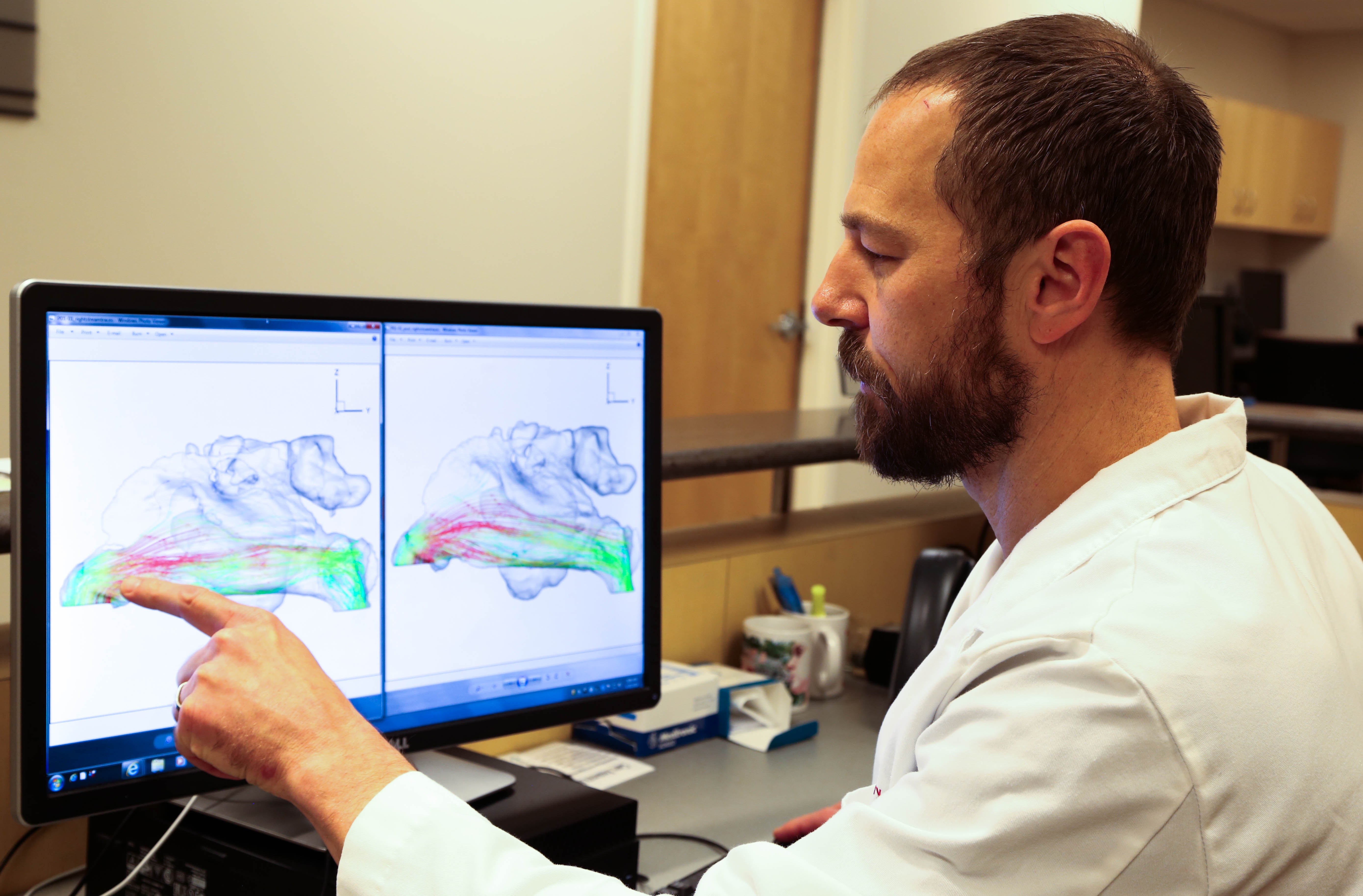 CT scans of the nasal cavity are taken before and after the procedure to measure airflow through the nasal cavity. This allows researchers to objectively measure changes in airflow.
CT scans of the nasal cavity are taken before and after the procedure to measure airflow through the nasal cavity. This allows researchers to objectively measure changes in airflow.“We use a technique called computational fluid dynamics, which is a study that we can base on CT scans that are done to show how the airflow through the nose travels,” said Otto. “Part of the goal of this study is to understand better how this technology changes the airflow through the nose to make people feel happy with their nasal breathing.”
Nasal obstruction can lead to chronic nasal congestion, difficulty breathing through the nose, difficulty sleeping and fatigue. Traditional treatments include medication, breathing strips and surgery to remove tissue and bone. Vivaer Nasal Airway Remodeling is performed in the doctor’s office under local anesthesia so patients can return to normal activities right away.
The study is recruiting patients ages 18 to 75 with chronic nasal obstruction due to the shape of the nasal value who have experienced positive response to temporary measures to open the nasal cavity such as with nasal strips and stents and where steroid medication failed to help. Patients with chronic sinusitis, prior nasal value surgery or severe septal deviation or polyps are among those excluded.
For more information about the clinical trial, visit the Ohio State Wexner Medical Center Division of Otolaryngology website. The study is being sponsored by Aerin Medical, the medical device company that developed the Vivaer Nasal Airway Remodeling Device.
###
